Congenital glaucoma - External and Internal Eye
Alerts and Notices
Important News & Links
Synopsis

Congenital glaucoma, distinguished primarily into primary congenital glaucoma (PCG) and secondary glaucoma, is a rare collection of pressure-sensitive ocular neuropathies brought about by ocular anatomical abnormalities. Given the risk of amblyopia and rapid permanent loss of the fragile pediatric visual system, early recognition and treatment of congenital glaucoma is paramount.
In PCG, abnormal neural crest cell migration results an isolated trabecular dysgenesis with impaired aqueous outflow. Specifically, irregularities in iris development and incorrect attachments to the trabecular meshwork thwart the flow of the aqueous into an otherwise normal canal of Schlemm.
Secondary infantile glaucoma occurs when aqueous outflow is abnormal due to a congenital or acquired ocular or systemic disorder, including aphakia, Axenfeld-Rieger syndrome, Sturge-Weber syndrome, Klippel-Trenaunay-Weber syndrome, uveitis, aniridia, and others.
The incidence of PCG is 1:10 000 to 20 000 live births in Western countries and as high as 1:2500 in the Middle East. Established studies on Middle Eastern populations with higher rates of PCG revealed a mutation in a cytochrome P450 CYP1B1 gene that metabolizes a signaling molecule involved in ocular development. In sporadic cases, 147 different mutations have been reported with significant heterogeneity worldwide.
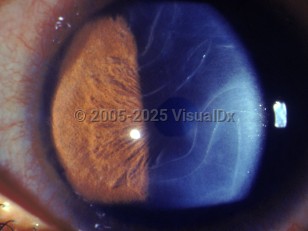
Classic symptoms include megalocornea, buphthalmos, corneal edema, and epiphora; the variable expression of phenotype results in a large diversity of presentation. In one large study of nearly 100 pediatric glaucoma patients, general practitioners and pediatricians were responsible for an equal number of referrals to the treating pediatric ophthalmologists at the tertiary eye care center as general community ophthalmologists. The mean age of presentation was 11 months. Examination under anesthesia was necessary to diagnose 75% of cases.
Eighty percent of affected children develop signs of glaucoma within the first year of life, and up to a quarter of patients demonstrate symptoms at birth. The syndrome occurs more frequently in males than females and is bilateral in 70%-80% of cases, although it may be asymmetric in onset. In general, later age of diagnosis implies less severe structural abnormalities and less optic nerve damage.
Without control of intraocular pressure (IOP), blindness due to optic nerve destruction and amblyopia is virtually guaranteed. While medical therapy can be used as a temporizing measure, surgery is the definitive treatment modality for the control of IOP in PCG. Medical treatment is the primary modality in secondary glaucoma, though many IOP-lowering medications must be used with caution in children. If the cornea is clear, the most commonly performed procedure for PCG is goniotomy. If the cornea is cloudy, trabeculotomy is the initial procedure of choice. Other surgical options such as trabeculectomy can be tried if these procedures fail.
In PCG, abnormal neural crest cell migration results an isolated trabecular dysgenesis with impaired aqueous outflow. Specifically, irregularities in iris development and incorrect attachments to the trabecular meshwork thwart the flow of the aqueous into an otherwise normal canal of Schlemm.
Secondary infantile glaucoma occurs when aqueous outflow is abnormal due to a congenital or acquired ocular or systemic disorder, including aphakia, Axenfeld-Rieger syndrome, Sturge-Weber syndrome, Klippel-Trenaunay-Weber syndrome, uveitis, aniridia, and others.
The incidence of PCG is 1:10 000 to 20 000 live births in Western countries and as high as 1:2500 in the Middle East. Established studies on Middle Eastern populations with higher rates of PCG revealed a mutation in a cytochrome P450 CYP1B1 gene that metabolizes a signaling molecule involved in ocular development. In sporadic cases, 147 different mutations have been reported with significant heterogeneity worldwide.
Classic symptoms include megalocornea, buphthalmos, corneal edema, and epiphora; the variable expression of phenotype results in a large diversity of presentation. In one large study of nearly 100 pediatric glaucoma patients, general practitioners and pediatricians were responsible for an equal number of referrals to the treating pediatric ophthalmologists at the tertiary eye care center as general community ophthalmologists. The mean age of presentation was 11 months. Examination under anesthesia was necessary to diagnose 75% of cases.
Eighty percent of affected children develop signs of glaucoma within the first year of life, and up to a quarter of patients demonstrate symptoms at birth. The syndrome occurs more frequently in males than females and is bilateral in 70%-80% of cases, although it may be asymmetric in onset. In general, later age of diagnosis implies less severe structural abnormalities and less optic nerve damage.
Without control of intraocular pressure (IOP), blindness due to optic nerve destruction and amblyopia is virtually guaranteed. While medical therapy can be used as a temporizing measure, surgery is the definitive treatment modality for the control of IOP in PCG. Medical treatment is the primary modality in secondary glaucoma, though many IOP-lowering medications must be used with caution in children. If the cornea is clear, the most commonly performed procedure for PCG is goniotomy. If the cornea is cloudy, trabeculotomy is the initial procedure of choice. Other surgical options such as trabeculectomy can be tried if these procedures fail.
Codes
ICD10CM:
Q15.0 – Congenital glaucoma
SNOMEDCT:
204113001 – Congenital glaucoma
Q15.0 – Congenital glaucoma
SNOMEDCT:
204113001 – Congenital glaucoma
Look For
Subscription Required
Diagnostic Pearls
Subscription Required
Differential Diagnosis & Pitfalls

To perform a comparison, select diagnoses from the classic differential
Subscription Required
Best Tests
Subscription Required
Management Pearls
Subscription Required
Therapy
Subscription Required
References
Subscription Required
Last Updated:03/18/2015
Congenital glaucoma - External and Internal Eye