Potentially life-threatening emergency
Inhalational anthrax - Chem-Bio-Rad Suspicion
See also in: OverviewAlerts and Notices
Important News & Links
Synopsis

Inhalational anthrax (historically known as "woolsorter's disease") is one of 4 major syndromes caused by Bacillus anthracis, an encapsulated, gram-positive, spore-forming bacillus. The other "forms" are cutaneous, gastrointestinal, and injection anthrax.
Naturally occurring cases of the inhalational form are extremely rare in developed countries. Bacillus anthracis has been classified by the Centers for Disease Control and Prevention (CDC) as a Category A bioterrorism agent, and if anthrax were to be intentionally dispersed, the most likely syndromes would be inhalational and cutaneous.
A single case of inhalational anthrax requires immediate notification of public health authorities because it may be due to criminal activity and may be a harbinger of subsequent cases. The incubation period for inhalational anthrax is usually 1-13 days and, rarely, up to 60 days. Hence, if the person presents with symptoms and has showered and/or changed their clothes since the exposure, the patient will not transmit disease to health care workers, since person-to-person transmission of inhalational anthrax does not occur.
Bacillus anthracis is present in both domestic and wild animals throughout the world (mainly in agricultural regions of South and Central America, Southern and Eastern Europe, Asia, Africa, the Caribbean, and the Middle East) and can be transmitted by their meat, wool, or hides. Therefore, veterinarians and those in the meat-, wool-, or hide-processing industries are the most at risk for contracting naturally occurring inhalational anthrax. Anthrax is rarely found in animals in the United States. An anthrax-like illness caused by Bacillus cereus group bacteria containing anthrax toxin genes has rarely been reported in welders and other metal workers in the United States.
In the past, there were considered to be 2 stages of anthrax: early and late. Based on the clinical, radiologic, and microbiologic findings of the 11 inhalational anthrax victims in 2001, 3 stages have been identified.
Early-prodromal:
The first stage consists of nonspecific symptoms such as fever, dyspnea, cough, headache, vomiting, chills, weakness, abdominal pain, and chest pain. Laboratory tests are also nonspecific at this stage. If diagnosed in the early prodrome stage, the prognosis is very good if the appropriate antibiotics are instituted. These initial symptoms occur for 24-48 hours, and the patient may show signs of transient improvement after 2-4 days.
Intermediate-progressive:
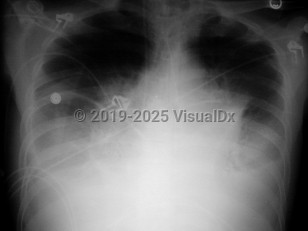
Inclusion criteria can be any one of the following: positive blood cultures, mediastinal adenopathy, or pleural effusions. The patient may also have nonspecific findings such as high fever, dyspnea, confusion, syncope, and nausea / vomiting. If diagnosed in this stage, the prognosis is still good if antibiotics are administered, pleural fluid is drained, and supportive care is provided.
Late-fulminant:
Inclusion criteria can be one of the following: meningitis, respiratory failure (requiring mechanical ventilation), or shock. Findings from the previous stages may be concurrently present. In 2001, no patient who required mechanical ventilation and/or hemodynamic support survived. More recently, a critically ill patient who was exposed unintentionally did survive with critical care support.
Much of anthrax morbidity and mortality is believed to be due to exotoxins. Source control with drainage of toxin reservoirs (eg, early, complete pleural drainage) and inhibition of additional toxin production (antibiotics) and toxin binding are mainstays of therapy for symptomatic patients.
Naturally occurring cases of the inhalational form are extremely rare in developed countries. Bacillus anthracis has been classified by the Centers for Disease Control and Prevention (CDC) as a Category A bioterrorism agent, and if anthrax were to be intentionally dispersed, the most likely syndromes would be inhalational and cutaneous.
A single case of inhalational anthrax requires immediate notification of public health authorities because it may be due to criminal activity and may be a harbinger of subsequent cases. The incubation period for inhalational anthrax is usually 1-13 days and, rarely, up to 60 days. Hence, if the person presents with symptoms and has showered and/or changed their clothes since the exposure, the patient will not transmit disease to health care workers, since person-to-person transmission of inhalational anthrax does not occur.
Bacillus anthracis is present in both domestic and wild animals throughout the world (mainly in agricultural regions of South and Central America, Southern and Eastern Europe, Asia, Africa, the Caribbean, and the Middle East) and can be transmitted by their meat, wool, or hides. Therefore, veterinarians and those in the meat-, wool-, or hide-processing industries are the most at risk for contracting naturally occurring inhalational anthrax. Anthrax is rarely found in animals in the United States. An anthrax-like illness caused by Bacillus cereus group bacteria containing anthrax toxin genes has rarely been reported in welders and other metal workers in the United States.
In the past, there were considered to be 2 stages of anthrax: early and late. Based on the clinical, radiologic, and microbiologic findings of the 11 inhalational anthrax victims in 2001, 3 stages have been identified.
Early-prodromal:
The first stage consists of nonspecific symptoms such as fever, dyspnea, cough, headache, vomiting, chills, weakness, abdominal pain, and chest pain. Laboratory tests are also nonspecific at this stage. If diagnosed in the early prodrome stage, the prognosis is very good if the appropriate antibiotics are instituted. These initial symptoms occur for 24-48 hours, and the patient may show signs of transient improvement after 2-4 days.
Intermediate-progressive:
Inclusion criteria can be any one of the following: positive blood cultures, mediastinal adenopathy, or pleural effusions. The patient may also have nonspecific findings such as high fever, dyspnea, confusion, syncope, and nausea / vomiting. If diagnosed in this stage, the prognosis is still good if antibiotics are administered, pleural fluid is drained, and supportive care is provided.
Late-fulminant:
Inclusion criteria can be one of the following: meningitis, respiratory failure (requiring mechanical ventilation), or shock. Findings from the previous stages may be concurrently present. In 2001, no patient who required mechanical ventilation and/or hemodynamic support survived. More recently, a critically ill patient who was exposed unintentionally did survive with critical care support.
Much of anthrax morbidity and mortality is believed to be due to exotoxins. Source control with drainage of toxin reservoirs (eg, early, complete pleural drainage) and inhibition of additional toxin production (antibiotics) and toxin binding are mainstays of therapy for symptomatic patients.
Codes
ICD10CM:
A22.1 – Pulmonary anthrax
SNOMEDCT:
11389007 – Inhalational anthrax
A22.1 – Pulmonary anthrax
SNOMEDCT:
11389007 – Inhalational anthrax
Look For
Subscription Required
Diagnostic Pearls
Subscription Required
Differential Diagnosis & Pitfalls

To perform a comparison, select diagnoses from the classic differential
Subscription Required
Best Tests
Subscription Required
Management Pearls
Subscription Required
Therapy
Subscription Required
References
Subscription Required
Last Updated:09/15/2024
Potentially life-threatening emergency
Inhalational anthrax - Chem-Bio-Rad Suspicion
See also in: Overview