Variegate porphyria
Alerts and Notices
Important News & Links
Synopsis

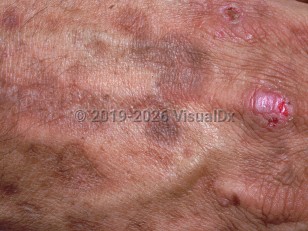
VP, also known as porphyria variegata, mixed porphyria, congenital cutaneous hepatic porphyria, and South African porphyria, is a blistering disorder caused by an autosomal dominantly inherited deficiency in protoporphyrinogen oxidase, a cytoplasmic enzyme involved in heme biosynthesis.
The prevalence is not known as many individuals are asymptomatic. Symptomatic VP has an estimated prevalence of 1:300 000 in Europe; this is higher in Southern Africans of Finnish and Dutch descent.
The spectrum of disease manifestation is broad, ranging from none to chronic photosensitivity and/or neurovisceral attacks. Acute neurovisceral attacks can be life-threatening. Homozygotes and compound heterozygotes tend to have more severe phenotypes.
The typical age of presentation is after puberty. Women are more commonly symptomatic for both skin and neurovisceral symptoms than men.
Cutaneous findings, which are the most common findings in VP, are caused by high plasma porphyrin levels and exposure of the skin to light with wavelengths in the range of 380-420 nm, with absorption peak at the Soret band, causing photoactivation, subsequent formation of reactive oxygen species, and tissue damage.
The exact pathogenesis of the acute attacks is poorly understood. It is thought that certain porphyrin precursors – such as ALA and porphobilinogen (PBG) – that are excreted by the liver in high amounts during attacks are potent neurotoxins.
Porphyrinogenic drugs (eg, barbiturates, phenytoin, other antiepileptics, rifampin), alcohol, smoking, hormonal changes, hormonal intake (eg progesterone), recurrent or chronic infections, gastric bypass surgery, and fasting or dieting (specifically carbohydrate restricting) may all precipitate an acute attack. Hepatic cytochrome p450 enzymes utilize heme as a cofactor; thus, medications degraded by the body through induction of these enzymes may accelerate heme synthesis and accumulation of the neurotoxic porphyrin precursors in these patients, thereby precipitating an acute attack. Such medications include barbiturates, estrogen / progesterone, griseofulvin, and sulfonamides, among others. The American Porphyria Foundation provides a Drug Safety Database Search that provides information about the interaction of specific drugs in patients with porphyria.
There is an increased risk of hepatocellular carcinoma in patients with VP, particularly those over 50 years of age; thus, routine monitoring is needed, particularly for those with other risk factors including cirrhosis, hepatitis C infection, or alcohol use disorder.
Codes
E80.20 – Unspecified porphyria
SNOMEDCT:
58275005 – Variegate porphyria
Look For
Subscription Required
Diagnostic Pearls
Subscription Required
Differential Diagnosis & Pitfalls

Subscription Required
Best Tests
Subscription Required
Management Pearls
Subscription Required
Therapy
Subscription Required
Drug Reaction Data
Subscription Required
References
Subscription Required
Last Updated:01/25/2022